|
BLUEBIRD - Fuel Cell & Battery Cartridge Technology
PLEASE USE OUR A TO Z INDEX TO NAVIGATE THIS SITE
|
|||
|
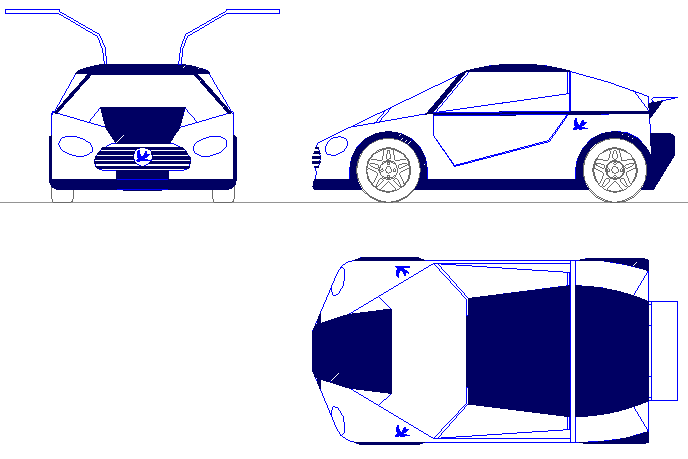
The Bluebird™ cartridge exchange system is suitable for use with fuel cells and/or batteries. This feature is considered to be the versatility EV's need that is missing at the moment, and is one of the reasons that there are no fuel cell cars in mass production at the moment - although, that is the plan. So, why not hedge your bets? In the circumstances it seems crazy not to.
Rather, we put it down to a lack of forward planning. With fuel cells still way off the mark in economic terms, a car that can swap between the two technologies at the flick of a switch, seems to us to be just the ticket.
PATENT SYSTEM - A comprehensive series of patents are being prepared in connection with the above system. In addition to patent protection designs copyright and trademarks will follow. These protections will protect collaborative partners as and when venture capital is needed to move to trials.
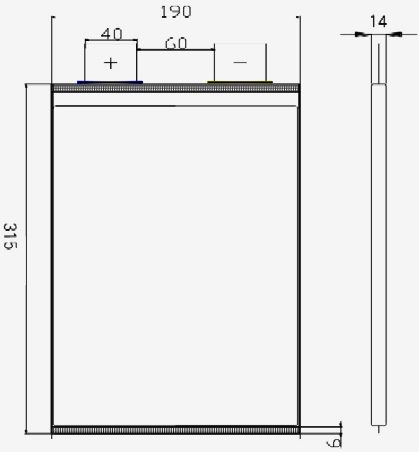
CARTRIDGE SIZE & CAPACITY
The Ecostar DC50™ 20Kw/hr cartridge will have the same connectors and loading (Bluebird) system but will be smaller as it has to be shoehorned into a more compact area than would be usual in any production EV that we might propose. The cartridge in this care will be 2400 x 300 x 240mm (94.5 X 11.75 X 9.5"), within which 28 x 3.6v x 200Ah lithium polymer cells will fit with ease (or similar). These cells measure 280 x 182 x 71mm and weigh 5.6Kg each. Thus our cartridge when using lithium battery technology will weigh: 5.6 x 28 = 156.8Kg + the weight of the cartridge and cabling (23+5Kg) give us 184.8Kg (407lbs). That's a whole lot less than when it was filled with lead-acid traction batteries for demonstrations.
Another example of a Bluebird™ smart-city "Universal" energy cartridge with 8 cubic feet capacity - so between 20-40kW hours of energy storage - depending on the chemistry used. This unit is suitable for a number of existing super-mini cars - and is compatible with the Bluebird™ service station prototype that is being proposed for a test town - such as in the Devonshire project. This cartridge may contain lithium batteries or fuel cell storage medium.
LOADING PHYSICS - MECHANICAL ADVANTAGE
Elementary my dear Watson. To lift a cartridge of this mass into position, a distance of some 150mm, will in theory require a .75Kw motor (or two motors to the equivalent wattage) operating for 0.5 of a second (now that is fast). The old formula for 1 horsepower is the ability to lift 550lbs one foot in one second. Translated to metric that is 249.5kg 300mm in one second (well above the 184.9Kg of our cartridge). One horsepower is equivalent to 746 watts. The beauty about electric horsepower, is that there is no manure.
In practical terms we don't want the weight of large electric motors onboard, except for the tractions motors, even though this is accepted practice in IC engined cars: in that they need a starter motor. Hydraulics is also a potential solution.
Two 24 volt motor examples, 250w direct drive 6mm chain sprocket and 450w with a 6.6:1 reduction box and 12mm chain sprocket.
It is much better to use 2 x 250w motors that would in theory be capable of lifting our cartridge 300mm in 1.5 seconds. Allow 60% for total losses in transmission takes us to 2.5 seconds lift time, the gearing for which should reduce a (sample) 2750 rpm output to 50 rpm at the output shaft (55:1 ratio) for a (sample) 180 degree rotation = lift sequence in 3 seconds (with reserve torque). Again being conservative, even though you might wish we were liberal and not labour the point.
The total amperage draw for a lift in 3 seconds @ 24 volts = 65A. A small lithium battery pack as commonly used by modelers would suffice for this duty, weighing in at 1Kg or less. The motors will weigh around 6Kg for inexpensive off the shelf items. you might agree that this weight penalty is well worth the ability to exchange cartridges using an onboard system. We can make the onboard system lighter still if we use smaller motors and wait for 6 seconds. At these sorts of recharging speeds, what's another few seconds? Of course, it's a different story for a club racer version of the Ecostar, when 3 seconds could cost you a race.
LITHIUM BATTERY COST
The bad news is that while lithium batteries are lighter than lead-acid, nickel-cadmium or nickel-metal hydride cells, they are expensive at around $260 per cell, making a cartridge $7,300 a go - or £4,334 at cost. That sounds frighteningly expensive.
The Ecostar city car promises the fastest battery cartridge exchanges - and it does it by itself with built in loaders - so reducing infrastructure costs significantly. Indeed, you don't need dedicated service stations to kickstart the system, just friendly garages who will have cartridges on their forecourts for the cars to pick up as they deposit their spent cartridge. This is roughly 2 times faster than the 2013 demonstration by Tesla, which was in itself twice as fast as filling a conventional petrol car, around 90 seconds.
THE COST OF EV MOTORING USING LITHIUM BATTERIES
Okay, so 7,300 / 2000 = $3.66 per recharge in depreciation (£2.17). Add to that the electricity to charge the pack (21 x £.14 = £2.94) and it costs us £5.11 per recharge to travel some 150 miles at daytime electricity tariffs, being ultra conservative in our estimations. That is the equivalent of 172 miles per gallon (where in the UK it is £5.87 a gallon of unleaded petrol) or 3.4 pence per mile (£0.034). Yup, that is three point four pence per mile, compared to 11.74 pence per mile (£0.1174) for a car that returns 50mpg.
Finally then, electric cars can compete with petrol cars on price and range. Now imagine service stations that re-charge replacement cartridges at off-peak prices. Add to that the higher price of servicing a petrol engine (oil changes, filters, spark plugs, etc) and there is no contest. Anyone who is economy minded must drive an EV.
Some of the most popular lithium iron phosphate batteries on the market. 3.6 volt cells and 200Ah and 180Ah capacities. The prices vary from one manufacturer and one supplier to another.
The ECOSTAR DC50 (BE4) as it began - is a two seat sports car project with front wheel drive and 50kW direct current (DC) electric motors. The roof will carry solar panels to supplement the lithium ion batteries or hydrogen fuel in cartridges as a range extender between instant energy transfer pit-stops. The cartridge may contain batteries, fuels cells, or a mix of batteries and fuel cells. The technology future proofs vehicles against battery and/or fuel cell advances - as an aid to the transition to a zero carbon lifestyle. The challenge for the ECOSTAR is to reduce the long standing 1 minute recharging world record, to less that 30 seconds. We know it is possible to get this down below 10 seconds, but our finances are running on empty, so we are aiming low. Watch this car turn into a motoring gem, as it rises like a Phoenix from the ashes. What color to paint it? There's no flies on us.
WHAT ABOUT FUELS CELLS?
Although there are currently no fuel cell vehicles available for commercial sale, over 20 fuel cell electric vehicle (FCEV) prototypes and demonstration cars have been released since 2009. Demonstration models include the Honda FCX Clarity, Toyota FCHV-adv, and Mercedes-Benz F-Cell. As of June 2011 demonstration FCEVs had driven more than 4,800,000 km (3,000,000 mi), with more than 27,000 refuelings. Demonstration fuel cell vehicles have been produced with "a driving range of more than 400 km (250 mi) between refueling". They can be refueled in less than 5 minutes.
The U.S. Department of Energy's Fuel Cell Technology Program claims that, as of 2011, fuel cells achieved 53–59% efficiency at one-quarter power and 42–53% vehicle efficiency at full power, and a durability of over 120,000 km (75,000 mi) with less than 10% degradation. In a Well-to-Wheels simulation analysis, that "did not address the economics and market constraints", General Motors and its partners estimated that per mile traveled, a fuel cell electric vehicle running on compressed gaseous hydrogen produced from natural gas could use about 40% less energy and emit 45% less greenhouse gasses than an internal combustion vehicle. A lead engineer from the Department of Energy whose team is testing fuel cell cars said in 2011 that the potential appeal is that "these are full-function vehicles with no limitations on range or refueling rate so they are a direct replacement for any vehicle. For instance, if you drive a full sized SUV and pull a boat up into the mountains, you can do that with this technology and you can't with current battery-only vehicles, which are more geared toward city driving."
HYDROGEN FUEL CELL CARTRIDGE - It looks like a battery cartridge. It performs like a battery cartridge. It stores energy like a lithium battery cartridge, but it is a hydrogen fuel store and fuel cell cartridge combination. Where lithium and cobalt raw materials may limit the production numbers of EVs as green motoring becomes the norm, hydrogen in a safe format might offer unlimited possibilities to resolve range anxiety. The concept is compatible with SMARTNET FASTCHARGE service stations. NOTE: This is just a concept, not a product yet. More development is needed to before mass production could be entertained. Design Copyright and patent(s) pending February 2020, all rights reserved Bluebird Marine Systems Ltd.
CAMBRIDGE UNIVERSITY - FAST CHARGE FIRES - MAY 2010
Scientists have identified a reason why lithium batteries in laptops and mobile phones may overheat and catch fire. Cambridge University researchers said the growth of metal fibres, called dendrites, could cause short circuits.
Nuclear Magnetic Resonance (NMR) spectroscopy, normally used to identify elements in molecules, has been adapted to "see" how the dendrites develop. Researchers said it could help solve fire safety problems which have held up the development of lithium batteries. Lithium battery technology is said to be crucial for progress on the next generation of electric cars.
When current
lithium batteries are charged quickly, minute lithium dendrites can form on carbon anodes.
These fibres can cause short circuits, causing the battery to rapidly overheat and catch fire, Professor Clare Grey, of Cambridge University's chemistry department
is quoted as saying: "These dead lithium fibres have been a significant impediment to the commercialisation of new generations of higher capacity batteries," "Fire safety must be solved before we can get to the next generation of lithium-ion batteries and before we can safely use these batteries in a wider range of transport applications. "Now that we can monitor dendrite formation inside batteries, we can identify when they are formed and under what conditions. "Our new method should allow researchers to identify which conditions lead to dendrite formation and to rapidly screen potential fixes to prevent the problem."
Cambridge University researchers monitoring fast charging that is causing lithium batteries to catch fire. Short circuits from dendrites are to blame.
LITHIUM BATTERY CONSTRUCTION
The three primary functional components of a lithium-ion battery are the anode, cathode, and electrolyte. The anode of a conventional lithium-ion cell is made from carbon, the cathode is a metal oxide, and the electrolyte is a lithium salt in an organic
solvent.
Li-ion cells are available in various formats, which can generally be divided into four
groups:
The lack of case gives pouch cells the highest energy density; however, pouch cells (and prismatic cells) require an external means of containment to prevent expansion when their state-of-charge (SOC) level is
high.
Varta lithium-ion battery, Museum Autovision, Altlussheim, Germany
Lithium batteries were first proposed by M.S.
Whittingham, now at Binghamton University, while working for Exxon in the
1970s. Whittingham used titanium(II) sulfide as the cathode and lithium metal as the anode.
Nissan Leaf's lithium-ion battery pack
In 1991, Sony and Asahi Kasei released the first commercial lithium-ion battery.
In 1996, Goodenough, Akshaya Padhi and coworkers identified lithium iron phosphate (LiFePO4) and other phospho-olivines (lithium metal phosphates with the same structure as mineral olivine) as cathode materials.
In 2002, Yet-Ming Chiang and his group at MIT showed a substantial improvement in the performance of lithium batteries by boosting the material's conductivity by doping it with aluminium, niobium and zirconium. The exact mechanism causing the increase became the subject of widespread
debate.
FUEL CELLS - FCH JTI PROGRAMME 2014
The Fuel Cells and Hydrogen (FCH) JTI is focused on securing long-term clean energy supplies for Europe in addition to the reduction of greenhouse gas emission from the energy and transport sectors.
A fuel cell is basically an electric chemical converter of fuel directly into electricity and heat, rather than traditional combustion. The fuel we are using is hydrogen, it is a very environmentally friendly gas. We try to improve the technology in both making hydrogen and converting hydrogen into cheaper, greener electricity – more efficiently.
According to the US Dept. of Energy, fuel cells will cost $30-$50 per kw-hr of output by 2017, depending on production volume. To put this number in perspective, Tesla battery packs are estimated to cost over $200 per kw-hr of output today and may fall to $140-175 per kw-hr by 2017. In all likelihood, fuel cell vehicles will cost less than battery electric vehicles by the end of the decade (barring some major decrease in battery costs, of course).
The estimated cost of building a hydrogen filling station is $3-5 million dollars, according to a report from the National Renewable Energy Laboratory. While that's definitely a lot of money, a standard gasoline filling station costs about $2 million to construct (according to the same NREL report).
The National Renewable Energy Laboratory has a functioning wind-powered hydrogen filling station in Boulder, Colorado that uses wind power to create hydrogen via electrolysis. German energy giant Linde will begin producing hydrogen at commercial scale via wind power by 2015. There are also solar panels in the research and testing phase that can break water down into hydrogen and oxygen via photoelectric synthesis and do so at extraordinary efficiency levels.
Battery electric vehicles have a low operating cost, and they plug in to an existing (and generally efficient) energy grid. However, most BEVs at this time can't be refueled in 3 minutes, and (as of now) can't go 300+ miles between refueling. BEVs aren't really feasible for use in pickup trucks and SUVs, at least not without serious compromises in tow and payload capacities and/or driving range.Fuel cell vehicles, on the other hand, have long range, fast refueling, and could easily be used in trucks or SUVs without sacrificing payload or tow capacity.
KEY
CHALLENGES
An
obvious solution to this chicken and egg situation is to make cars with
a universal cartridge, such as the Bluebird™ system. These cartridges
may contain fuel cells and storage tanks, rather than batteries. Thus, a
car that is purchased with a battery cartridge, may change to a fuel
cell cartridge at any dealership. Problem solved. HORIZON 2020 - TRANSPORT RESEARCH & INNOVATION - FCH2
The JTI was re-launched at the start of 2014 with extra funding and a focus on developing “clean, efficient and affordable fuel cells and hydrogen technologies”. It is important to invest in energy storage technology.
We are very happy to see
that European
Commission proposals are set to receive an important increase in our budget, with a focus on energy
storage – energy storage is something mankind has have never come to terms with and we have never found a good solution to store energy in large quantities. The problem is not the availability of the energy, it’s the
storage.”
ECOSTAR DC50 LINKS A-Z INDEX
EXTERNAL LINKS
http://www.horizonhydrogeneenergie.com/ http://www.fch-ju.eu/news/new-website-programme-horizon-hydrog%C3%A8ne-energie-h2e http://horizon2020projects.com/sc-transport-interviews/storing-energy-in-horizon-2020/ http://horizon2020projects.com/sc-transport-interviews/powering-the-future/ http://cordis.europa.eu/fp7/ideas/home_en.html http://cordis.europa.eu/fp7/ideas Parts for Toyota fuel cell myths https://parts.olathetoyota.com/fuel-cell-myths.html http://ec.europa.eu/programmes/horizon2020/en/ http://ec.europa.eu/transport/themes/research/horizon2020_en.htm http://www.agnimotors.com/site/ http://www.wired.com/autopia/2012/09/formula-e/ http://www.telegraph.co.uk/finance/The-electric-cars-of-the-future.html http://news.bbc.co.uk/1/hi/england/cambridgeshire/8687963.stm Rechargeable Li-Ion OEM Battery Products Panasonic Develops Higher-Capacity Li-Ion Cells; Application of Silicon-based Alloy in Anode The effect of PHEV and HEV duty cycles on battery and battery pack performance (PDF) Vapor-grown carbon fiber anode for cylindrical lithium batteries. Journal of Power Sources Battery Types and Characteristics for HEV ThermoAnalytics, Inc., 2007 Electrovaya, Tata Motors to make electric Indica Cleantech Group Mixed Conductors for Lithium Batteries. mrs.com; Materials Research Society MSDS: National Power Corp Lithium Ion Batteries (PDF). Tektronix Inc., 7 May 2004 Battery Management Systems for Large Lithium-Ion Battery Packs page 2 Cell boards for various cell formats". Elithion.com Battery Management Systems for Large Lithium-Ion Battery Packs page 234 "USPTO search for inventions by "Goodenough, John"". Patft.uspto.gov US 4304825, Basu; Samar, "Rechargeable battery", issued 8 December 1981, assigned to Bell Telephone Laboratories Gholamabbas Nazri, Gianfranco Pistoia (2004). Lithium batteries: science and ... - Google Books. Springer Voelcker, John (September 2007). Lithium Batteries Take to the Road IEEE Spectrum. US 4668595, Yoshino; Akira, "Secondary Battery", issued 10 May 1985, assigned to Asahi Kasei Phospho-olivines as positive-electrode materials for rechargeable lithium batteries". Electrochem. Society Editors (6 March 2008). "In search of the perfect battery" (PDF). The Economist Staff (November 2003) (PDF). Lithium Ion technical handbook. Gold Peak Industries Ltd. "Impedance Analysis of Silicon Nanowire Lithium Ion Battery Anodes" (PDF) C. K. Chan; X. F. Zhang, Y. Cui (2007). "High Capacity Li-ion Battery Anodes Using Ge Nanowires" (PDF) Liquid Electrolyte Systems for Advanced Lithium Batteries (PDF). Chem Eng Research Info Center "A123 M1 cell specifications". Battery Management Systems for Large Lithium-Ion Battery Packs page 12 Buchmann, Isidor (200804). "Choosing a battery that will last". Isidor Buchmann (CEO of Cadex Electronics) Battery Management Systems for Large Lithium-Ion Battery Packs page 229 Buchmann, Isidor (September 2006). "BatteryUniversity.com: How to prolong lithium-based batteries" Buchmann, Isidor (February 2003). "Advanced battery analyzers". Isidor Buchmann "Lithium-ion Battery Charging Basics". PowerStream Technologies AeroVironment achieves electric vehicle fast-charge milestone AeroVironment, 30 May 2007 "Charging Lithium-ion Batteries". batteryuniversity.com. "The 3 charging stages". Liionbms.com. http://liionbms.com/php/wp_charging_stages.php Battery Management Systems for Large Lithium Ion Battery Packs section 6.2.3 Kevin Jost [ed.] (October 2006). Tech Briefs: CPI takes new direction on Li-ion batteries (PDF) Voelcker, John (September 2007). Lithium Batteries Take to the Road. IEEE Spectrum. Loveday, Eric (23 April 2010). "Hitachi develops manganese cathode, could double life of li-ion batteries". Nikkei Nissan On Track Nickel Manganese Cobalt Li-ion Cell for 2015 Green Car Congress Bulkeley, William M. (26 November 2005). "New Type of Battery Offers Voltage Aplenty, at Premium" (2 November 2005) A123Systems Launches Higher-Power, Faster Recharging Green Car Congress. Imara Corporation website Imaracorp.com. http://www.imaracorp.com. Retrieved 8 October 2011. Battery Company Says Its Technology Boosts Hybrid Battery Performance Green Car Advisor; Edmunds Inc. A multifunctional 3.5 V iron-based phosphate cathode for rechargeable batteries A Research First: Lithium Air Battery Development (Press Release) 17 November 2009 Vanadium Modified LiFePO4 Cathode for Li-ion Batteries Acceptance of the First Grid-Scale, Battery Energy Storage System" (Press release) 21 November 2008 Marty Ozols (11 November 2009). Altair Nanotechnologies Power Partner - The Military Microsoft PowerPoint - 061125 Altair EDTA Presentation Altairnano.com. Blain, Loz (2 November 2007). "Subaru doubles the battery range on its electric car concept". gizmag. Li-Ion Rechargeable Batteries Made Safer Nikkei Electronics Asia. 29 January 2008. gatech.edu". http://www.mse.gatech.edu. Palca, Joe (6 April 2009). Hidden Ingredient In New, Greener Battery: A Virus. npr.org; National Public Radio. Zandonella, Catherine (11 April 2009). "Battery grown from "armour plated" viruses". New Scientist Bullis, Kevin (28 September 2006). "Powerful Batteries That Assemble Themselves". technologyreview Bad Virus Put to Good Use". Clark School of Engineering, University of Maryland. 6 December 2010. Self-Assembled Nanocomposites Boost Lithium-Ion Battery Anodes Nature Materials http://www.gatech.edu/newsroom/release.html?nid=54920. New Nanowire Battery Holds 10 Times The Charge Of Existing Ones". Science Daily. 20 December 2007. http://www.sciencedaily.com/releases/2007/12/071219103105.htm Interview with Dr. Cui, Inventor of Silicon Nanowire Lithium-ion Battery Breakthrough GM-Volt. Metal hydrides for lithium-ion batteries Nature Materials 7 (11): 916–921. 2008NatMa...7..916O Laboratory of the Prof. Gleb Yushin Nano-tech.gatech.edu. 31 August 2011. "Deformations in Si−Li Anodes Electrochemical Alloying in Nano-Confined Space American Chemical Society (30 September 2011). "A Simple Way to Boost Battery Storage". Technology Review Welcome to Ener1. Ener1 (Press release). Archived from the original 8 July 2006. EnerDel Technical Presentation (PDF). EnerDel Corporation. 29 October 2007. Bullis, Kevin (22 June 2006). Higher-Capacity Lithium-Ion Batteries Technology Review How to Prolong Lithium-based Batteries". Battery University Modelling Lithium-ion cells, Analysis Lithium-Ion Battery Degradation during Thermal Aging (PDF). http://www.electrochem.org/dl/ma/204/pdfs/0253.PDF About Battery Management Systems White Paper - CCCV chargers: a false sense of security. ELithion LLC. Commercial Power (9 September 2006). "Safety handling guidelines for Lithium Batteries" (PDF). http://marine.rutgers.edu "Safety Last". The New York Times. http://www.nytimes.com/2006/09/01/opinion/01cringely.html Nokia issues BL-5C battery warning, offers replacement. Wikinews. 14 August 2007. Staff (27 July 2007). Nokia N91 cell phone explodes Mukamo - Filipino News (blog). Dell Recalls Lithium Batteries". Chemical and Engineering News:11; American Chemical Society Dell laptop explodes at Japanese conference. The Inquirer. Kyocera Launches Precautionary Battery Recall" (Press release). Kyocera Wireless. 28 October 2004. "Safe Travel". Safetravel.dot.gov. U.S. Department of Transportation. 1 January 2008 U.S. Department of Transportation revises lithium battery rules press release". Little Guy Media. http://www.robgalbraith.com/bins/content_page.asp?cid=7-9206-9211 Prohibitions - 6.3.12 - Dangerous, offensive and indecent articles (PDF). Hong Kong. December 2009 International Mail > FAQs > Goods/Services: Shipping a Laptop Japan Post Service Co. Ltd. http://arstechnica.com/science/news/2011/08/new-solid-state-compound-lithium-ion-batteries.ars Melody Voth (6 December 2010). "Battery Booster". http://pubs.acs.org/cen/email/html/8849bus1.html What Are Batteries, Fuel Cells, and Supercapacitors?" (PDF). Chemical Review 104 (104): 4245. 10.1021/cr020730k. http://pubs.acs.org/doi/pdf/10.1021/cr020730k. Lithium batteries at the Open Directory Project The Future of Electric Vehicles on Lithium Availability Journal of Energy Security
BATTERY MANUFACTURERS
Tianjin CLZ technology co., ltd (Beijing office)
OSN Power Tech Limited
AGA Technology Co., Ltd F13,Zhantao Technology Building
Aga Technology offer a range of LiPo/NiMh for RC Models/Airsoft, and saddle packs with hardcases for cars,
LinYi Gelon LIB Co.,Ltd
Intelligent Battery Support System THE BLUE BIRDS OF HAPPINESS
|
|||
|
This
website is Copyright © 2014 Bluebird Marine Systems Limited.
The names Bluebird™,
Blueplanet BE3™, Ecostar
DC50™
and the blue bird in flight
|